 However, because of the resonance delocalization of the lone pair, it interconverts from sp 3 to sp 2 as it is the only way of having the electrons in an aligned p orbital that can overlap and participate in resonance stabilization with the pi bond electrons of the C=O double bond. The nitrogen atom here has steric number 4 and expected to sp 3. One exception with the steric number is, for example, the amides. Again, for the same reason, that its steric number is 3 ( sp 2 – three identical orbitals). However, the carbon in these type of carbocations is sp 2 hybridized. Here the carbon has only single bonds and it may look like it is supposed to be sp 3 hybridized. The other two 2p orbitals are used for making the double bonds on each side of the carbon.Īnother common, and very important example is the carbocations. This is only possible in the sp hybridization. There a few common exceptions to what we have discussed about determining the hybridization state and they are mostly related to the method where we look at the bonding type of the atom.įor example, in the carbon dioxide (CO 2), the carbon has two double bonds, but it is sp-hybridized.Īnd the reason for this is the fact that the steric number of the carbon is two (there are only two atoms of oxygen connected to it) and in order to keep two atoms at 180 o, which is the optimal geometry, the carbon needs to use two identical orbitals. 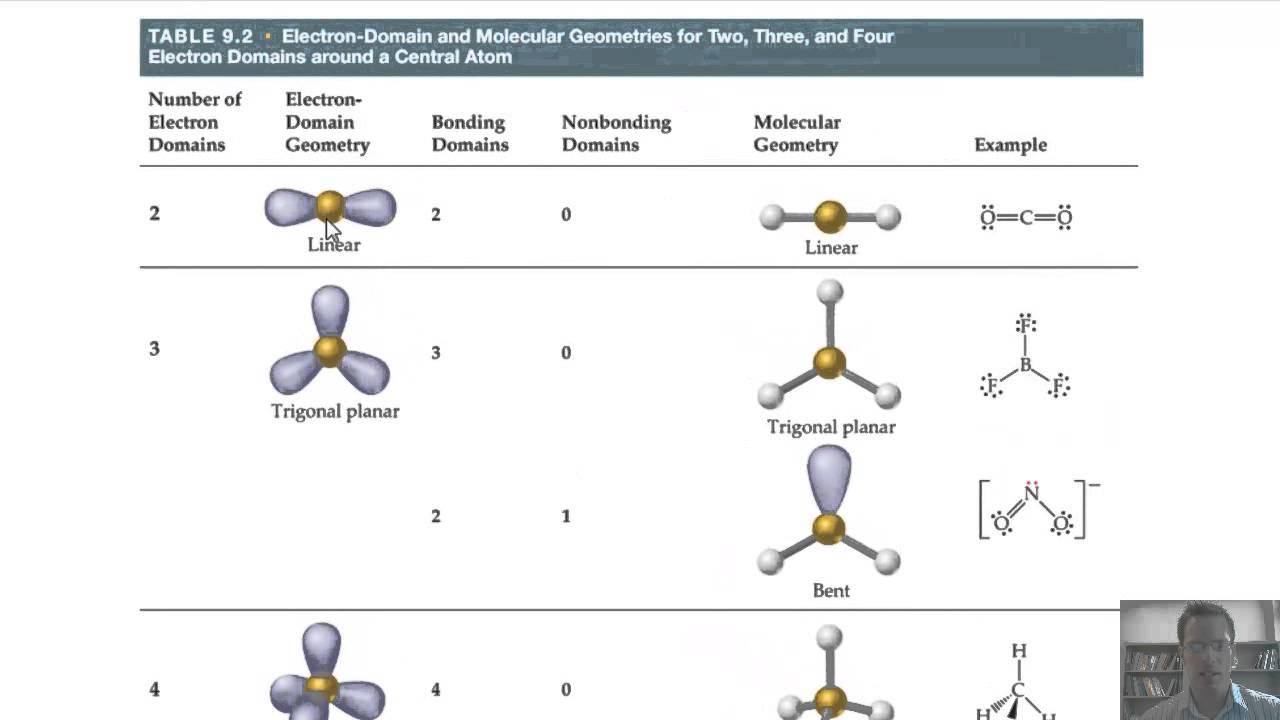 
The triple bond, on the other hand, is characteristic for alkynes where the carbon atoms are sp-hybridized. 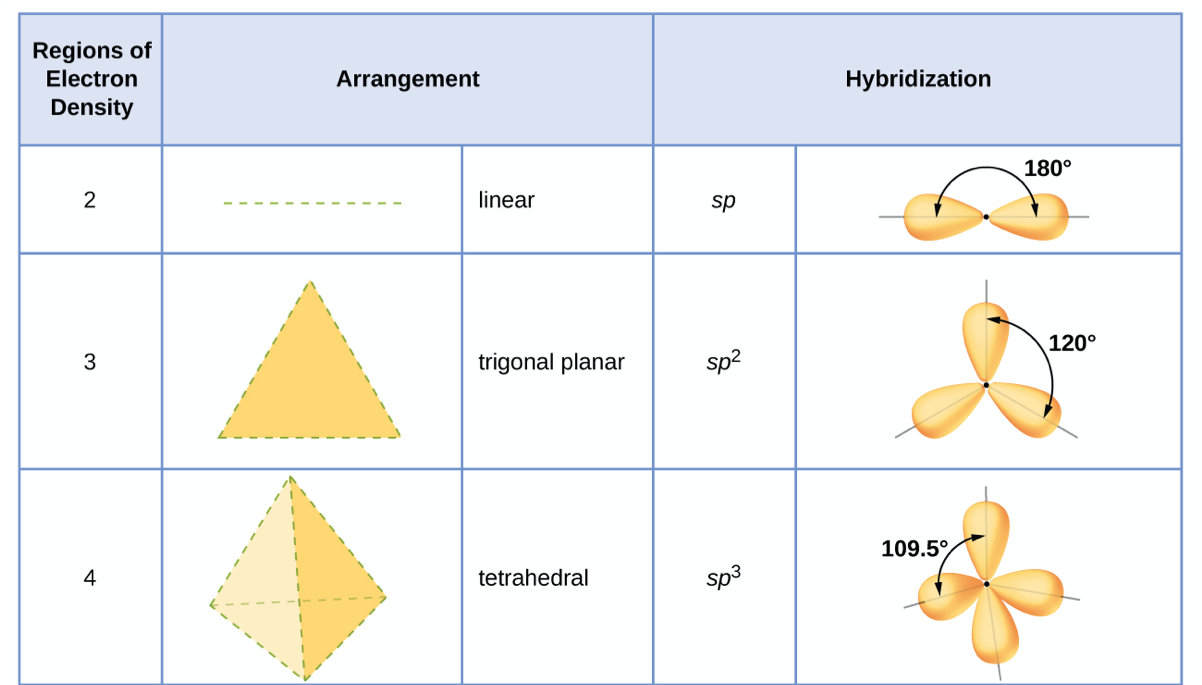
The carbons in alkenes and other atoms with a double bond are often sp 2 hybridized and have trigonal planar geometry. All the carbon atoms in an alkane are sp 3 hybridized with tetrahedral geometry. In general, an atom with all single bonds is an sp 3 hybridized. In addition to this method, it is also very useful to remember some traits related to the structure and hybridization. Other methods to determine the hybridization N8 – SN = 4(3 atoms + 1 lone pair), therefore it is sp 3Ĭ9 – SN = 2 (2 atoms), therefore it is spĬ10 – SN = 2 (2 atoms), therefore it is sp O5 – SN = 4(2 atoms + 2 lone pairs), therefore it is sp 3Ĭ6 – SN = 4 (4 atoms), therefore it is sp 3Ĭ7 – SN = 4(4 atoms), therefore it is sp 3 O4 – SN = 3 (1 atom + 2 lone pairs), therefore it is sp 2 So now, let’s go back to our molecule and determine the hybridization states for all the atoms.Ĭ1 – SN = 3 (three atoms connected), therefore it is sp 2Ĭ2 – SN = 3 (three atoms connected), therefore it is sp 2 Once you know how to determine the steric number (it is from the VSEPR theory), you simply need to apply the following correlation: Notice that multiple bonds do not matter, it is atoms + lone pairs for any bond type. This is also known as the Steric Number (SN).īelow are a few examples of steric numbers 2-4 which is largely what you need to know in organic chemistry: By groups, we mean either atoms or lone pairs of electrons. The first thing you need to do is determine the number of the groups that are on each atom. 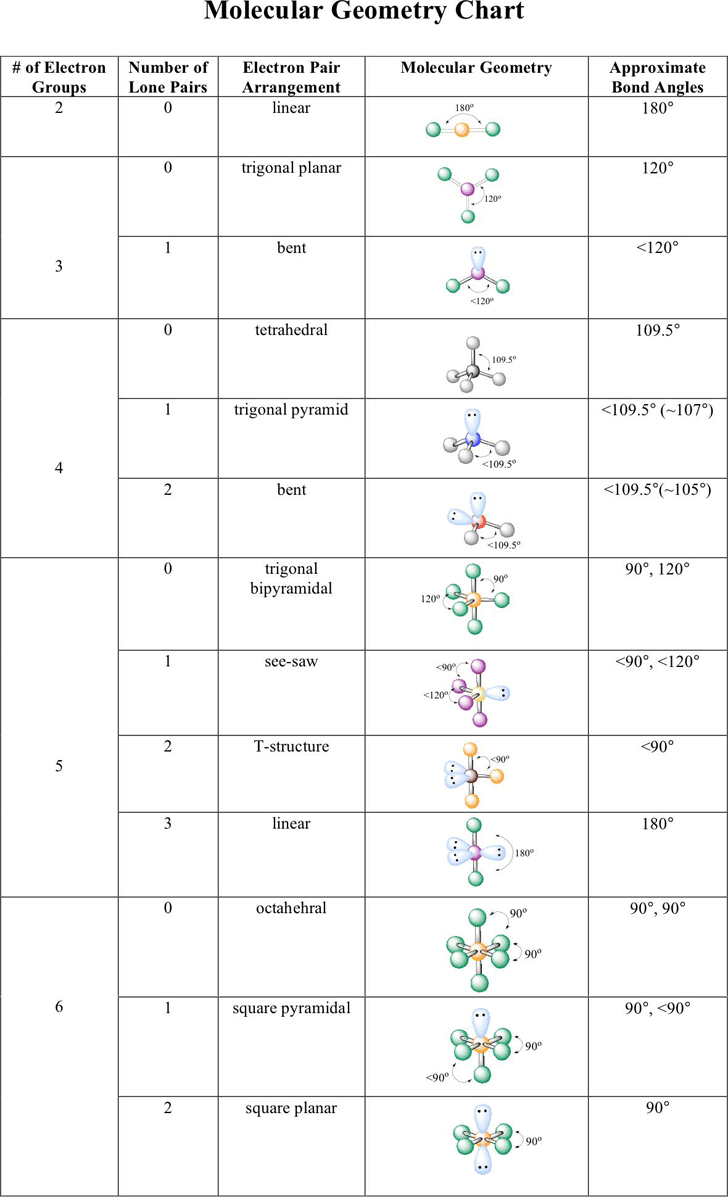
Let’s say you are asked to determine the hybridization state for the numbered atoms in the following molecule: The hybridization theory is often seen as a long and confusing concept and it is a handy skill to be able to quickly determine if the atom is sp 3, sp 2 or sp without having to go through all the details of how the hybridization had happened.įortunately, there is a shortcut in doing this and in this post, I will try to summarize this in a few distinct steps that you need to follow. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
